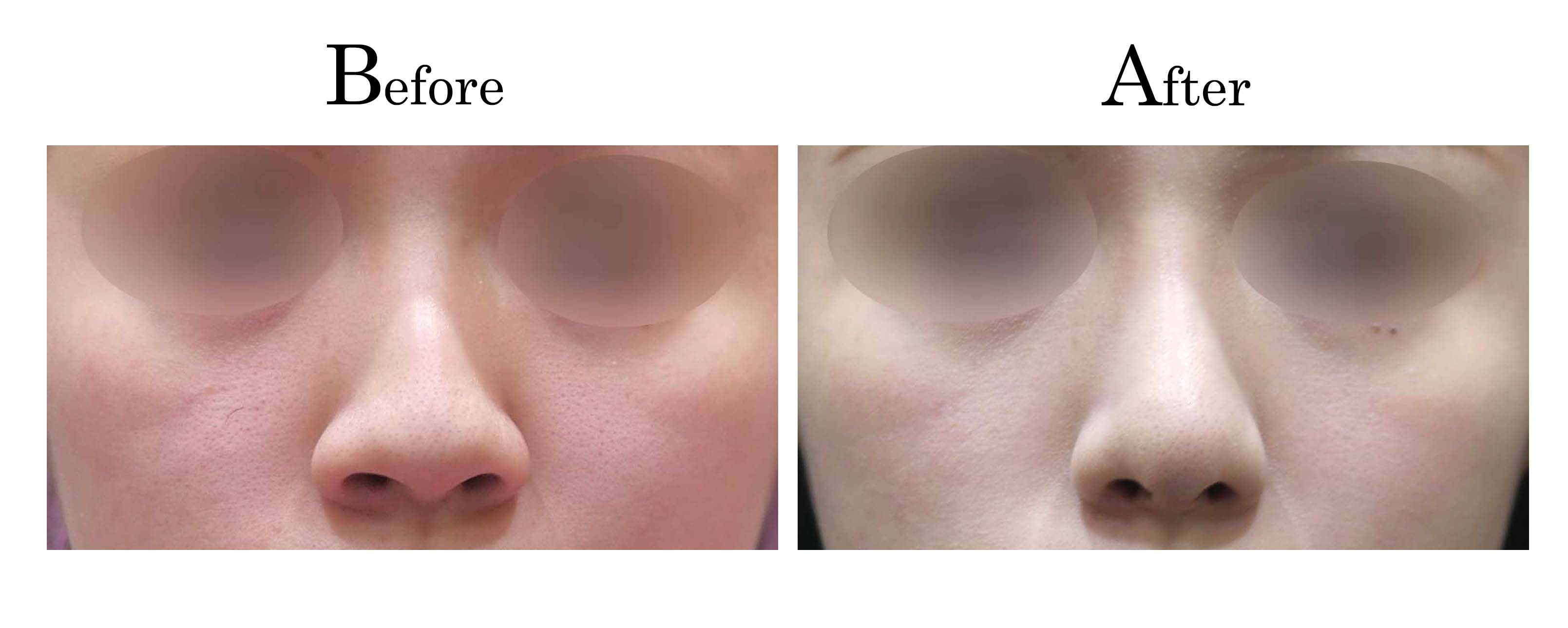
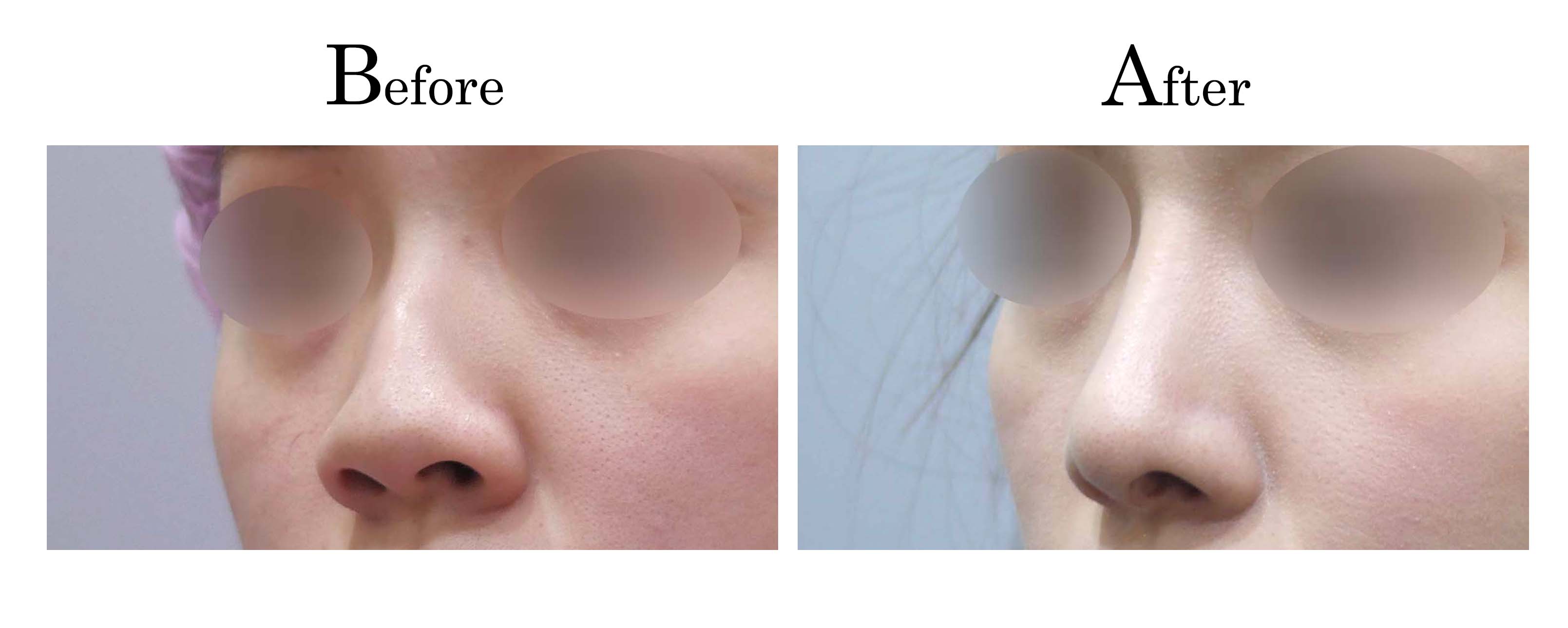
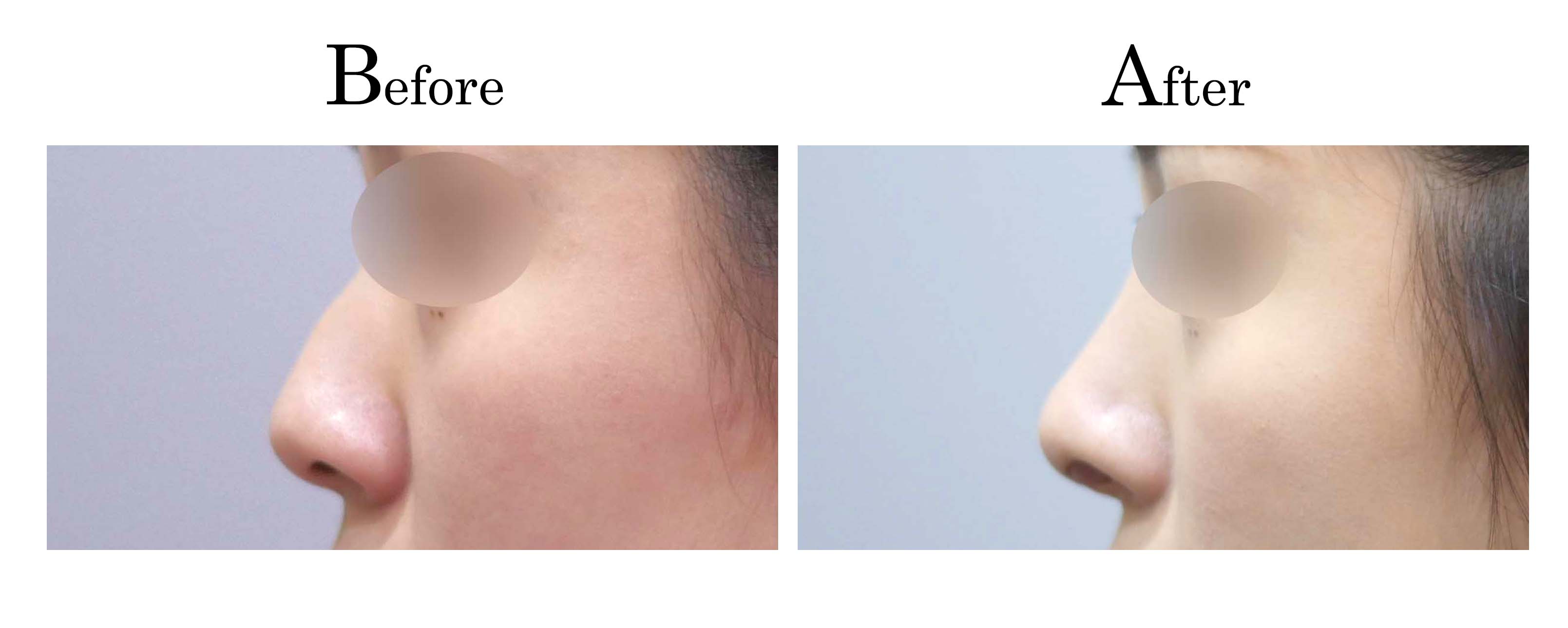
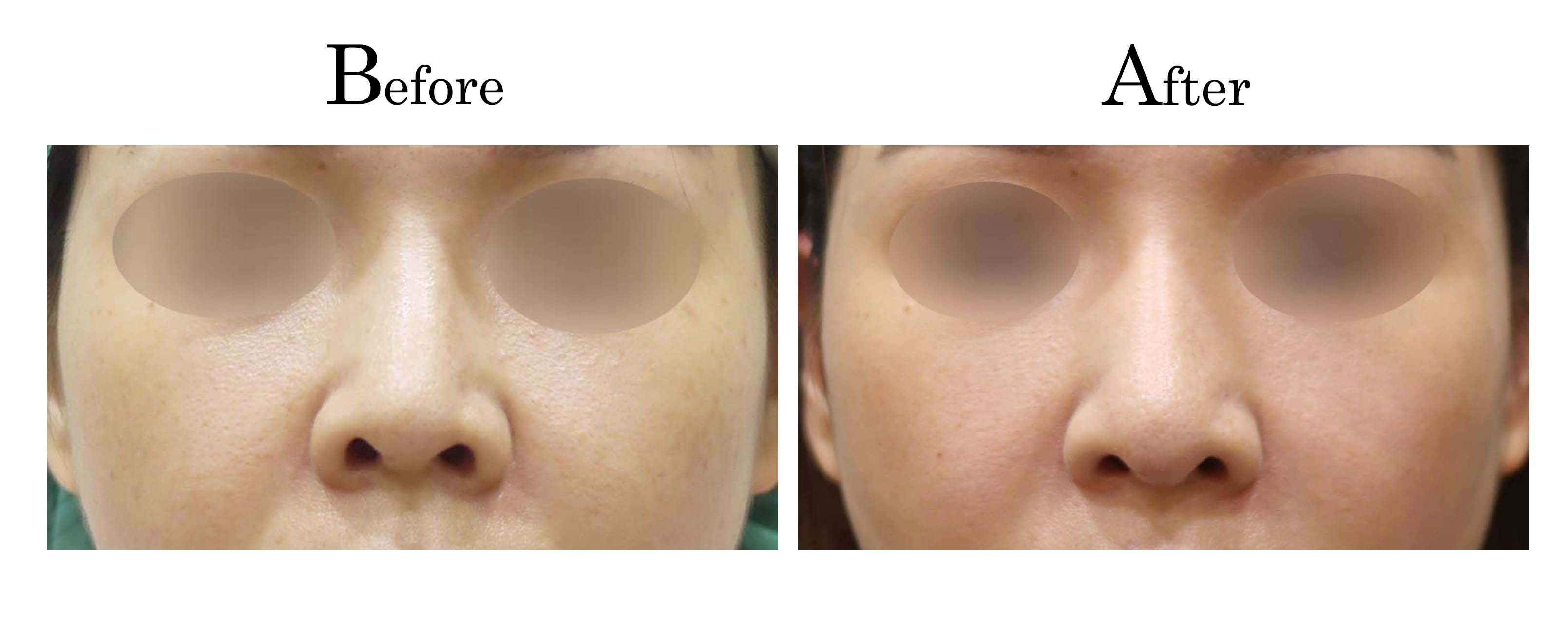
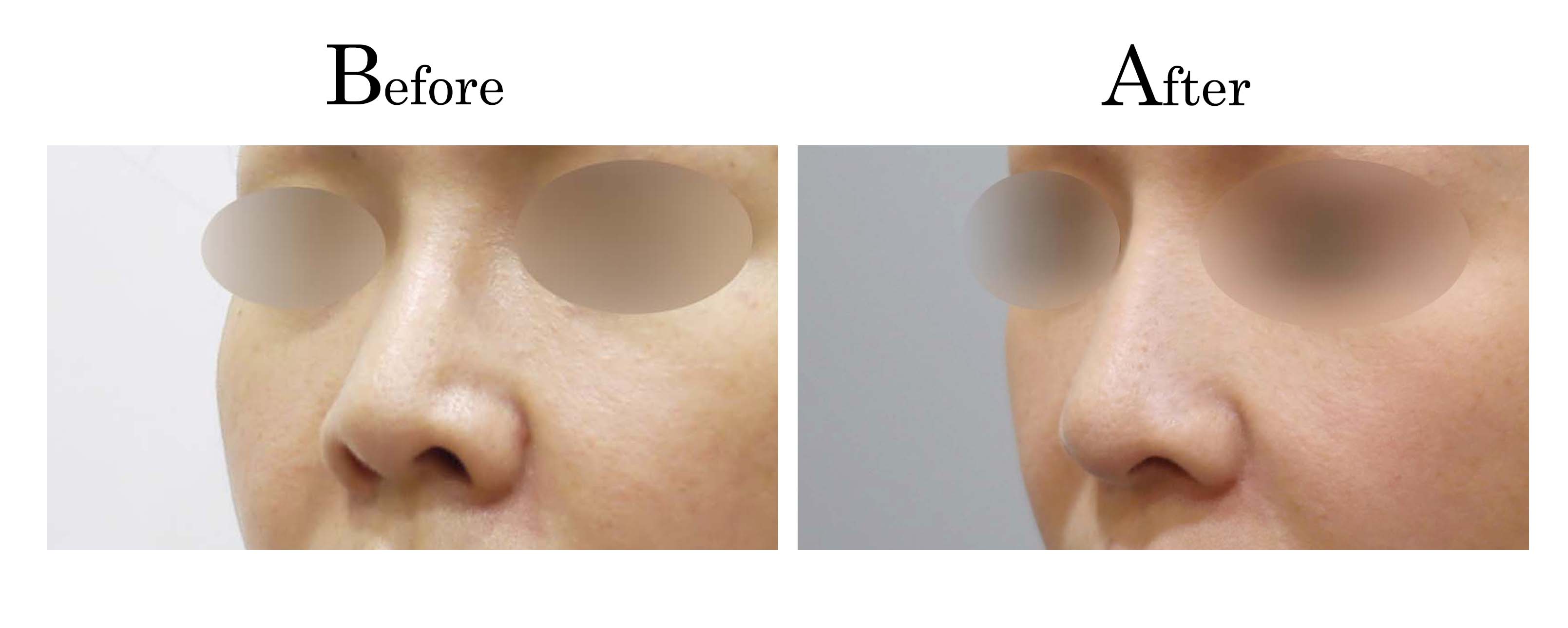
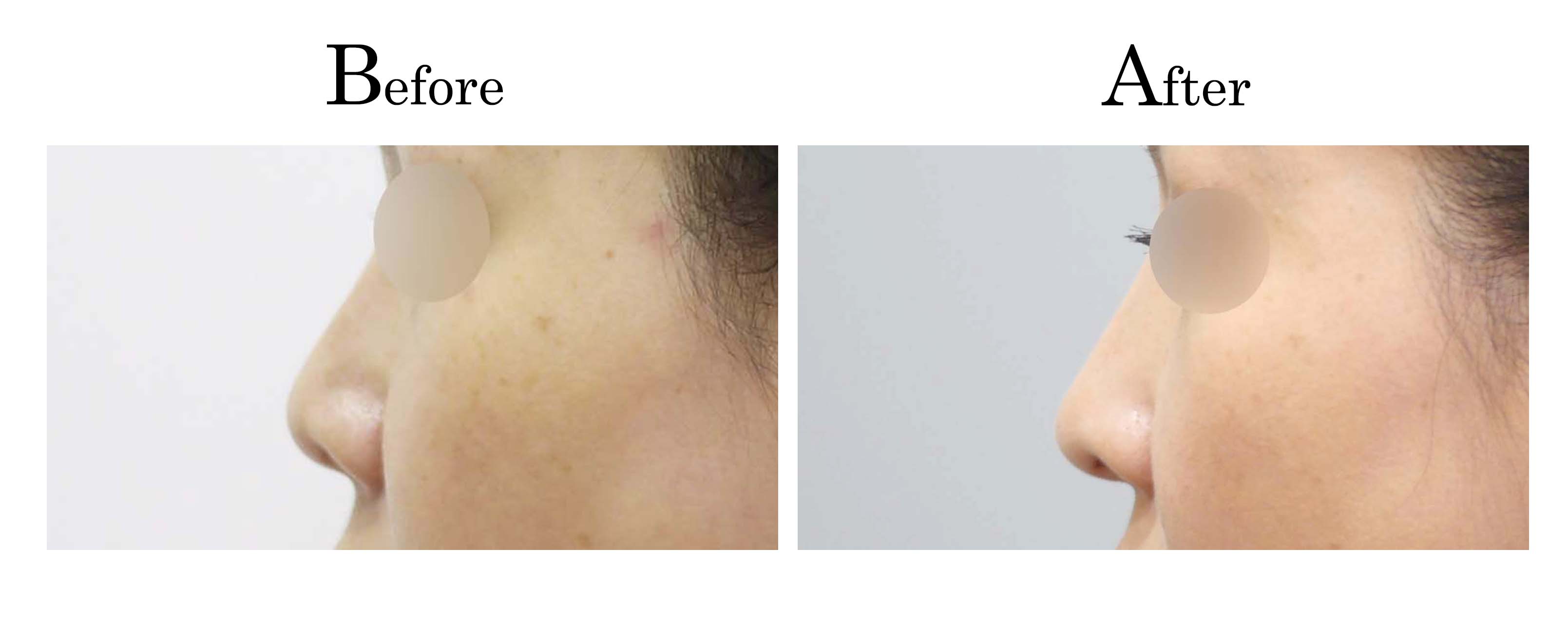
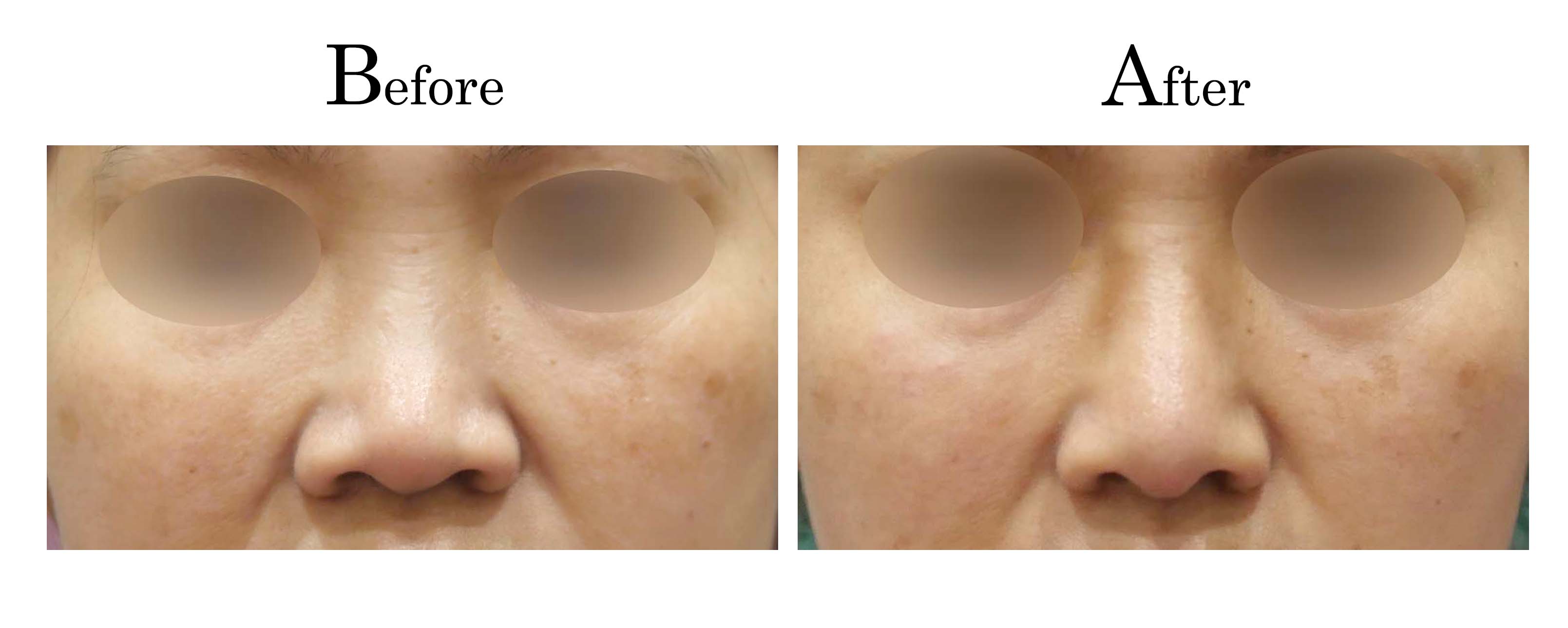
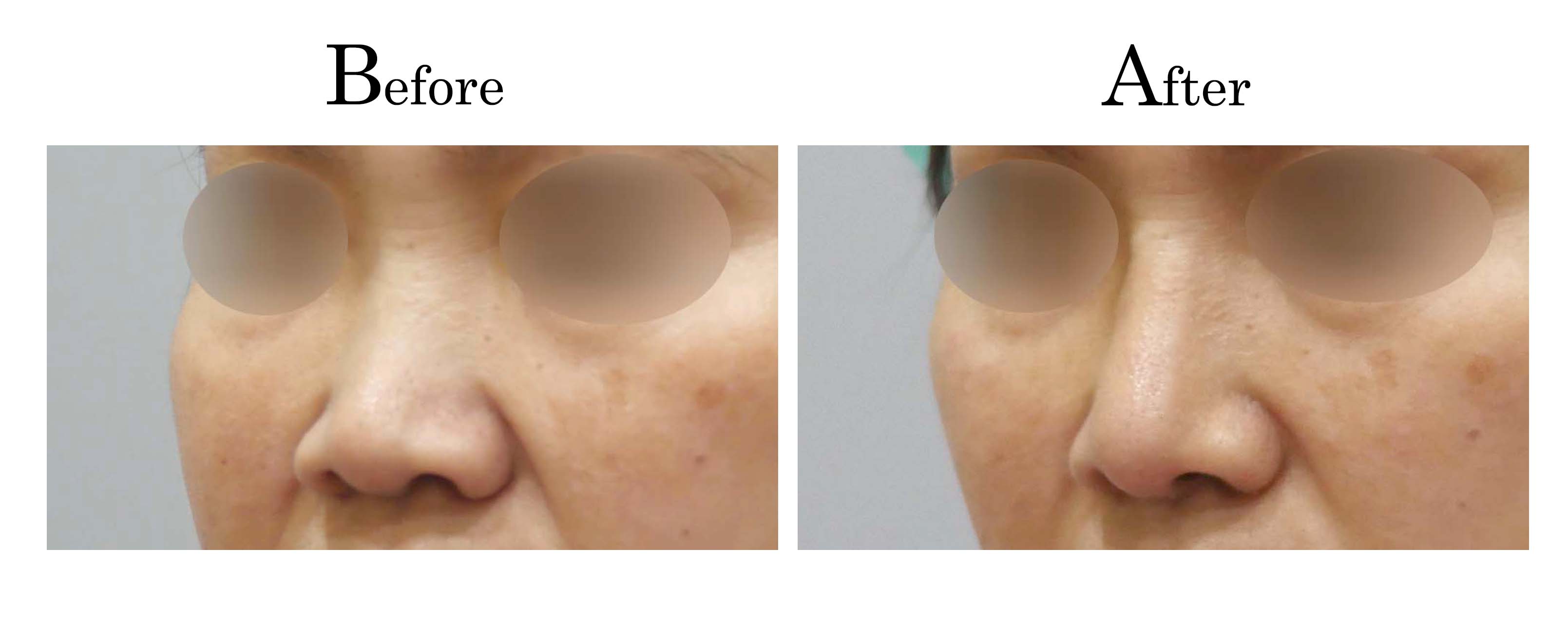
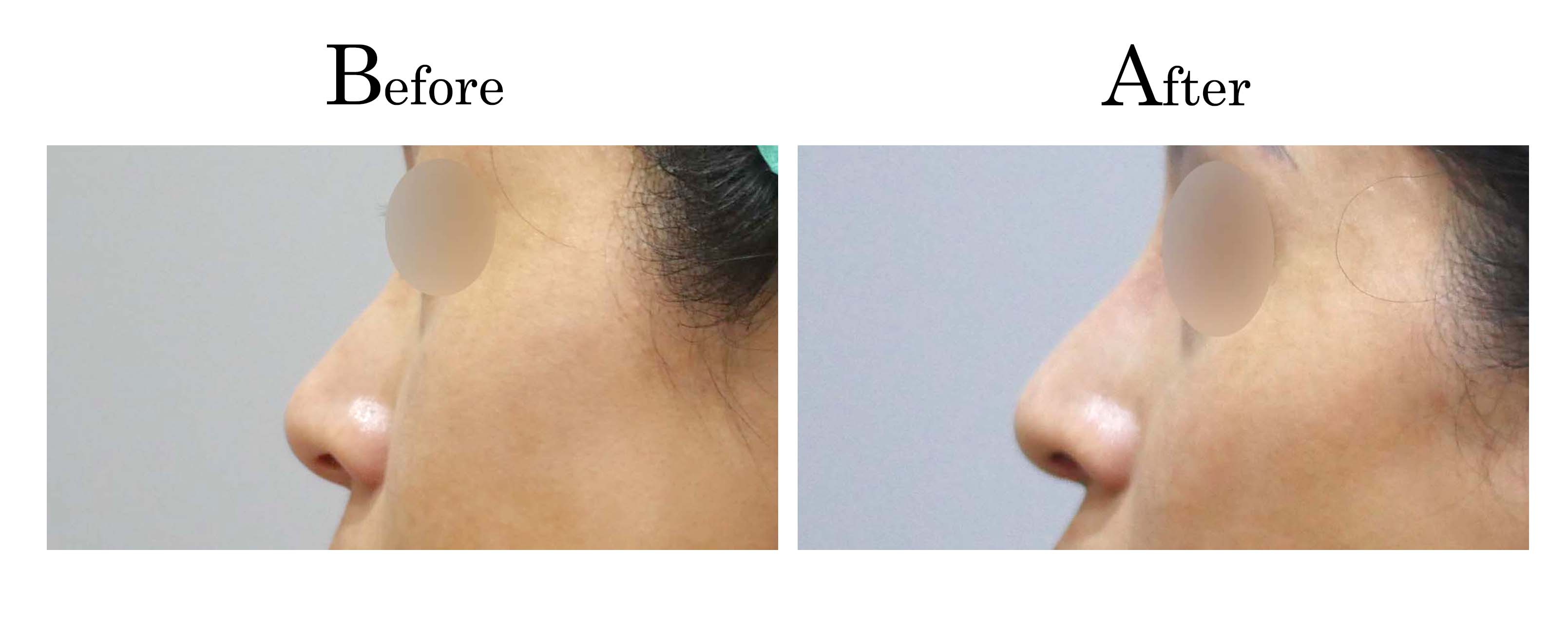
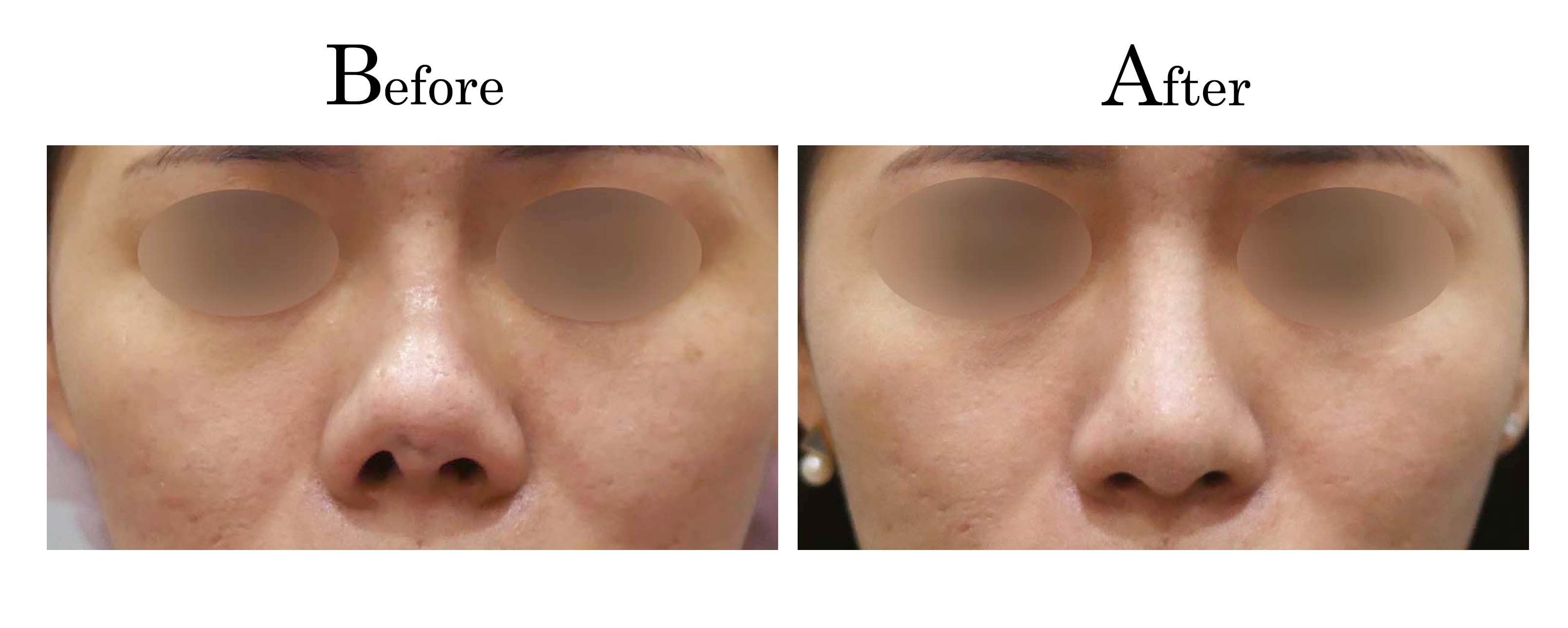
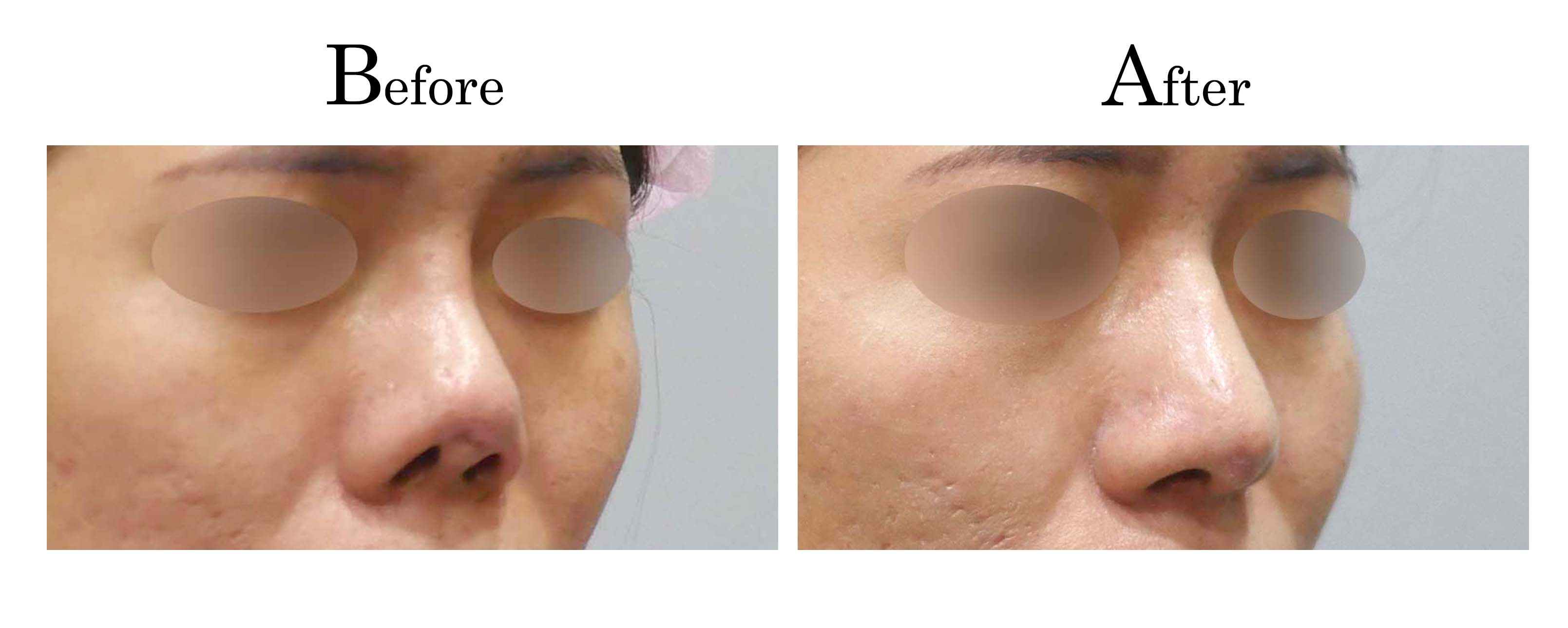
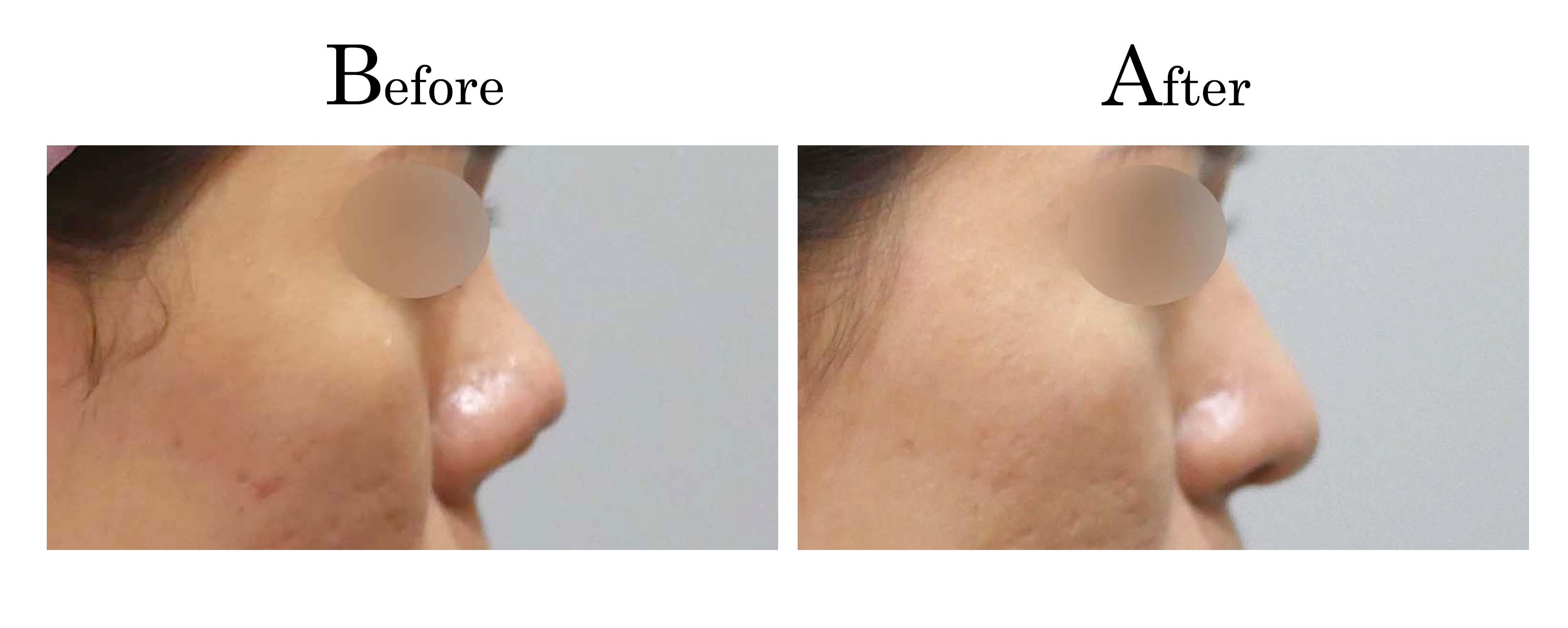
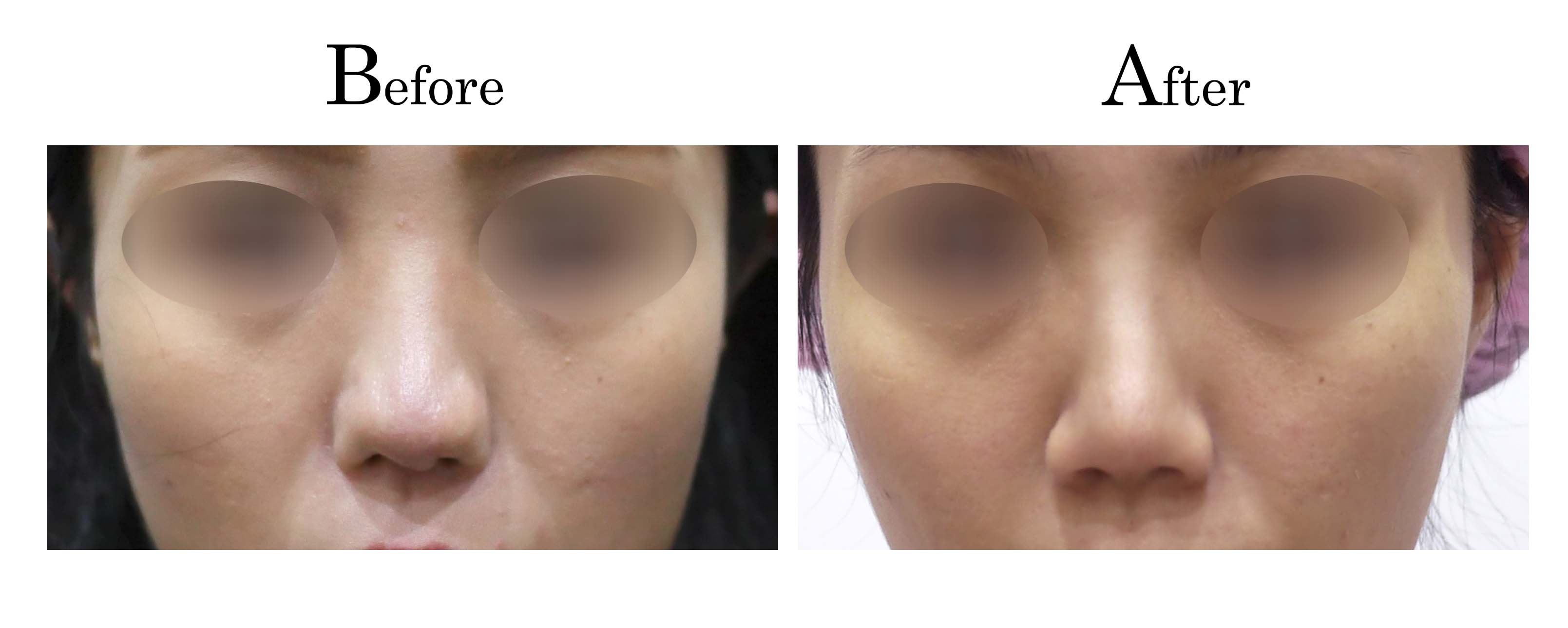
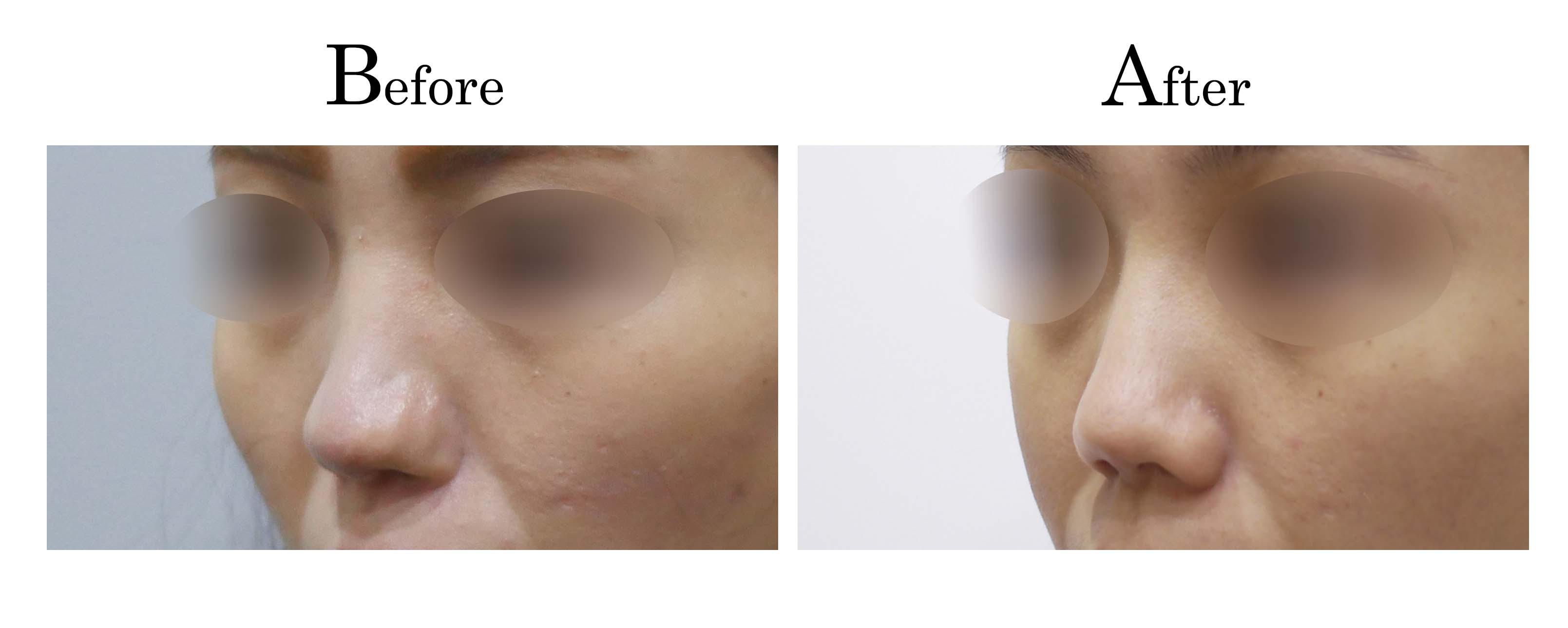
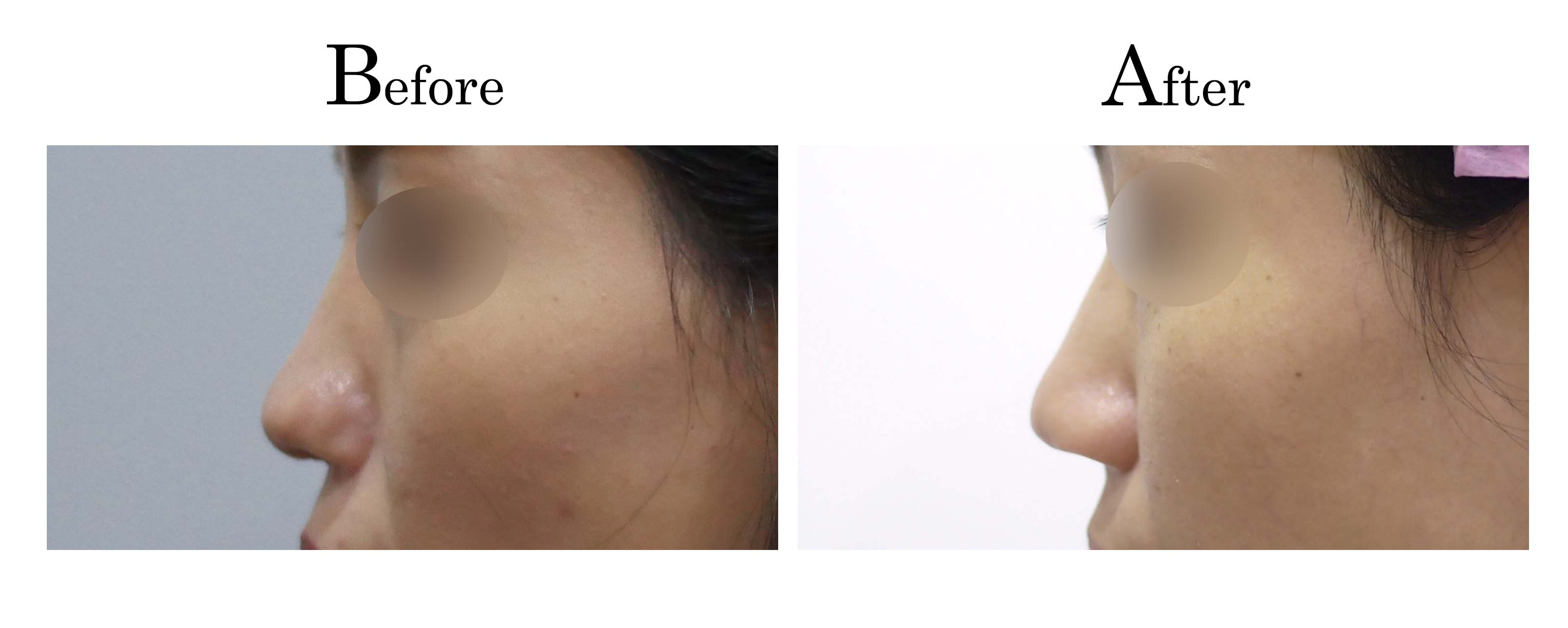
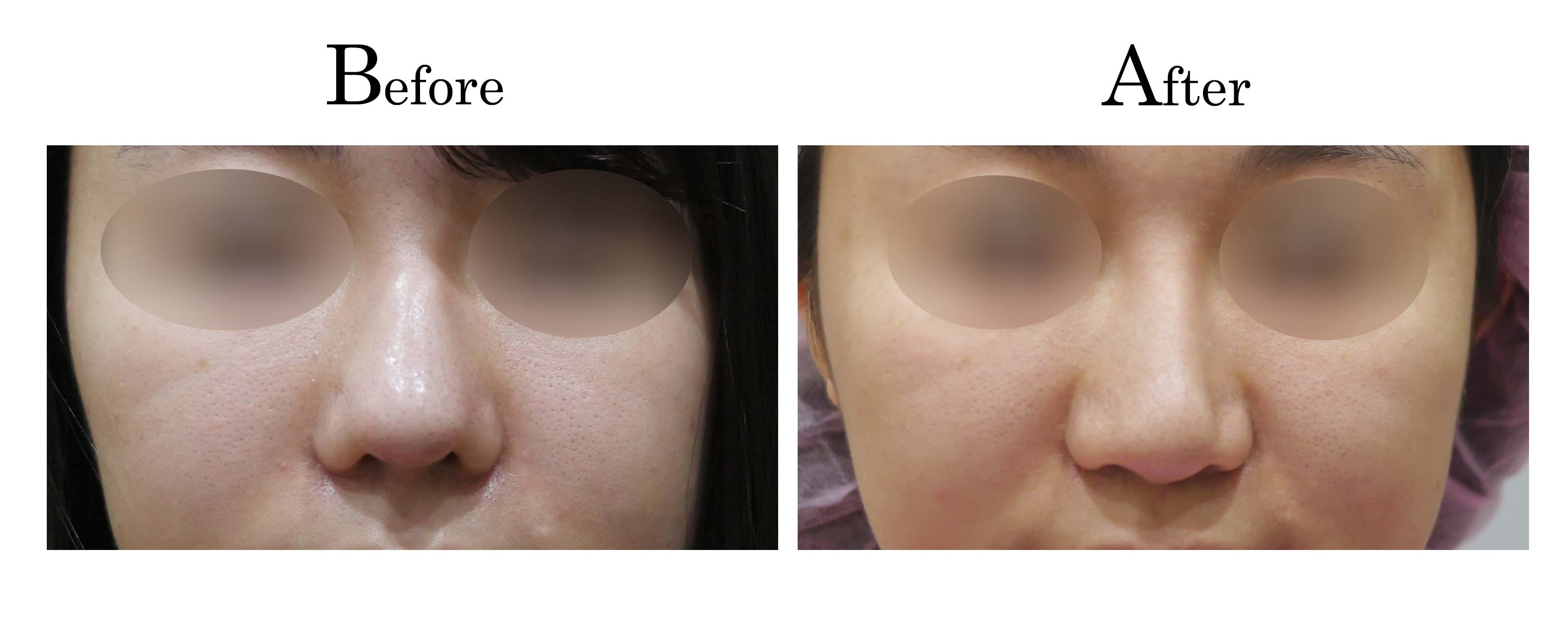
教主醫美整形外科診所<
您的位置在:首頁 > 醫療團隊
台北-邱昱勳醫師Team

邱醫師與各國醫師學術交流:


邱醫師上各大新聞醫學解說:
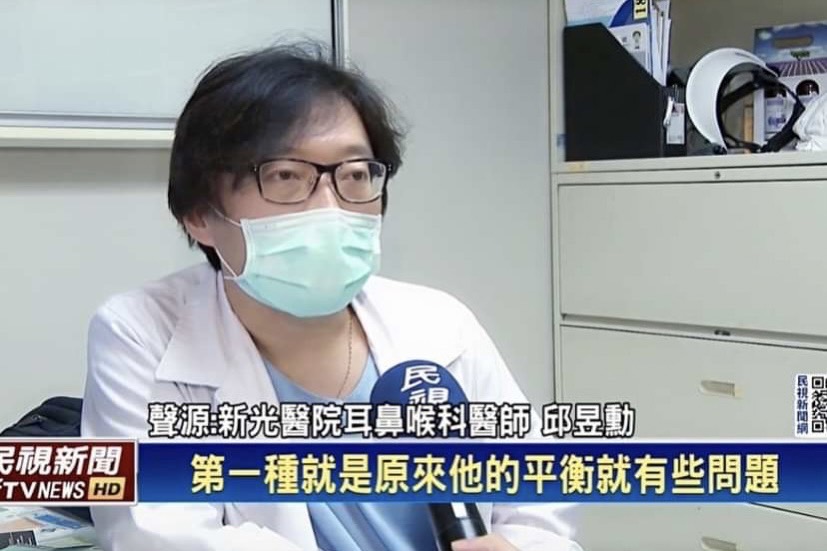
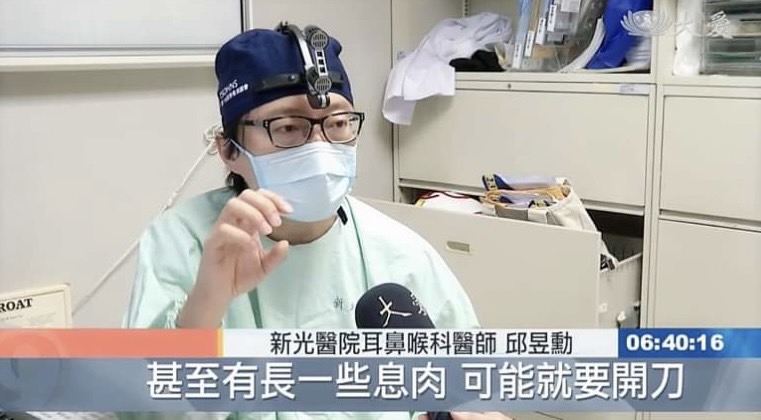
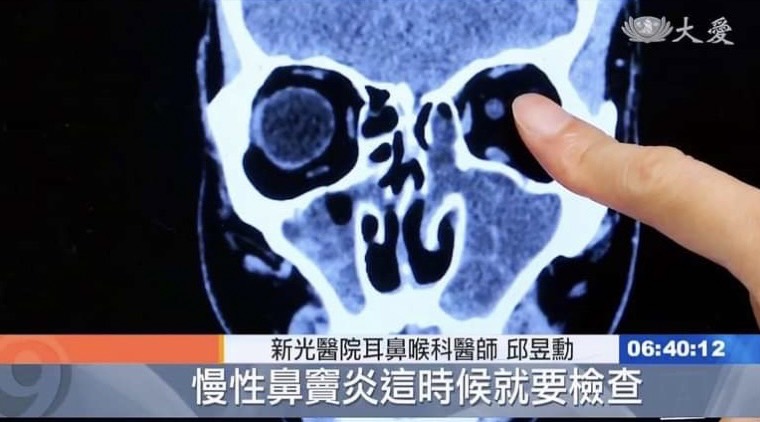
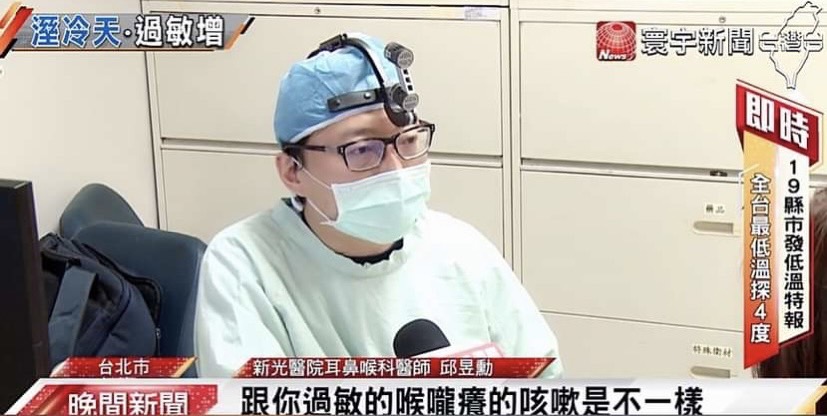

邱醫師到各國學術演講:










學經歷:
-世界五大洲鼻整形演講醫師
-臺大醫及工學院醫學工程研究所博士
-美國芝加哥大學醫學中心鼻整形手術研究
-中整協海峽兩岸分會常務委員
-鼻整形委員會副主任委員
-臺灣耳鼻喉頭頸外科醫學會
-顏面整形重建訓練委員會委員
-臺灣顏面整形重建外科醫學會理事
-國立台灣大學醫學院附設醫院耳鼻喉部教學主治醫師
-中國醫藥大學附設醫院臺北分院耳鼻喉科主任
-振興醫院醫美中心主治醫師
-泛亞顏面整形重建外科醫學會 (PAAFPRS) 台灣代表
-韓國KCCS 醫學雜誌 編輯醫師
-IFOS, AAFPRS,EAFPS, KCCS, AMWC, IMCAS, IFFPSS, PAAFPRS國際鼻整形講師
專長:
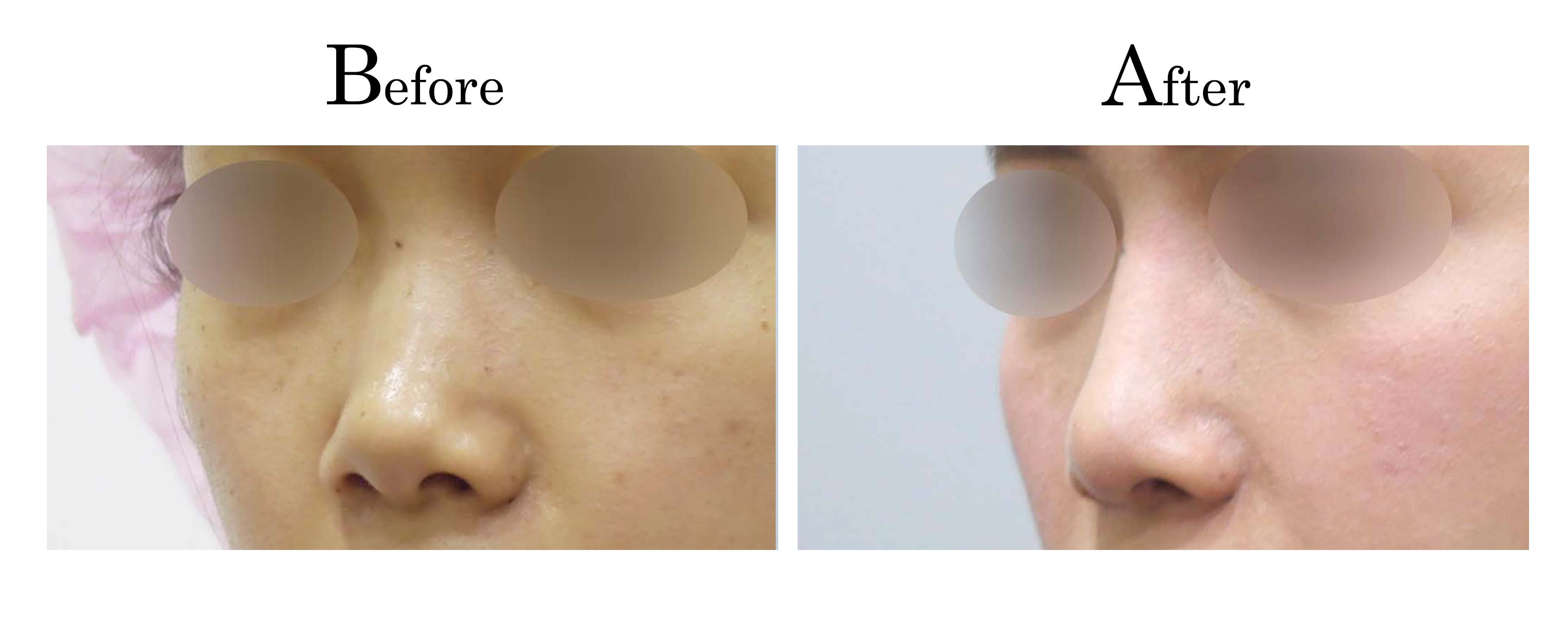
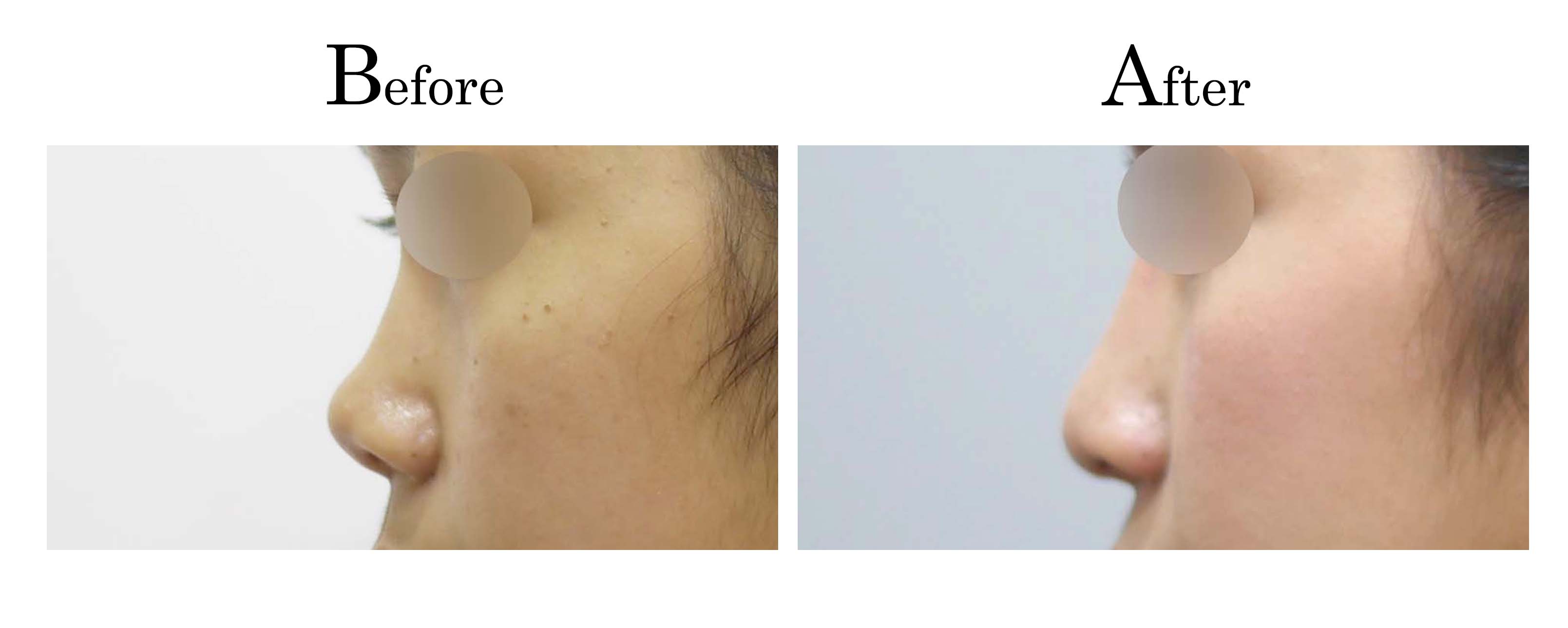
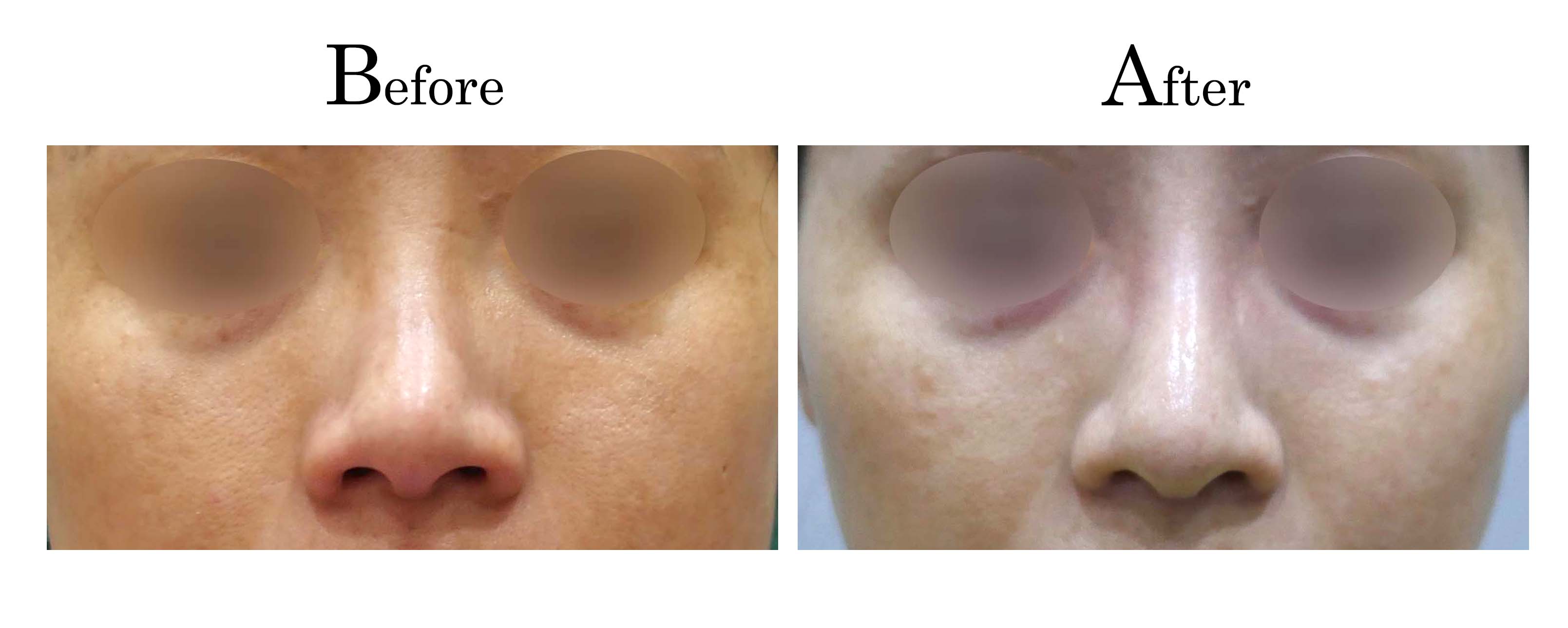
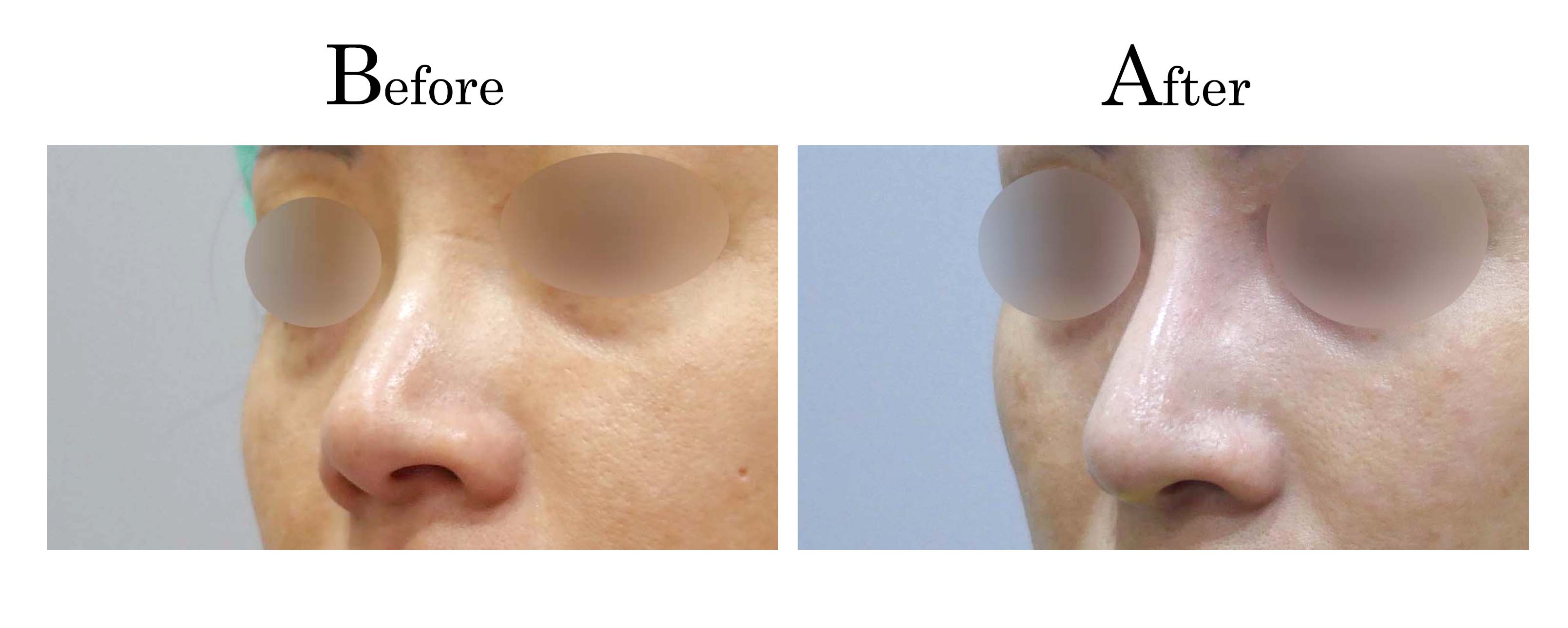
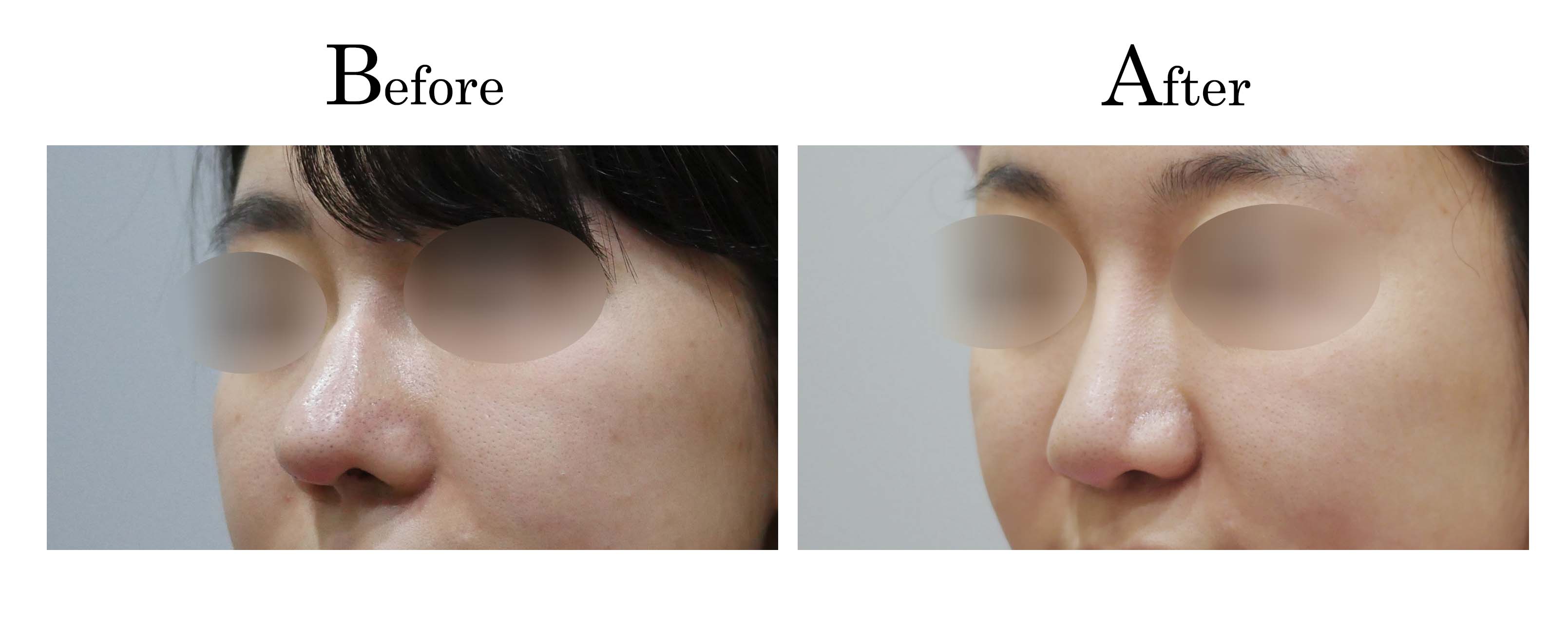
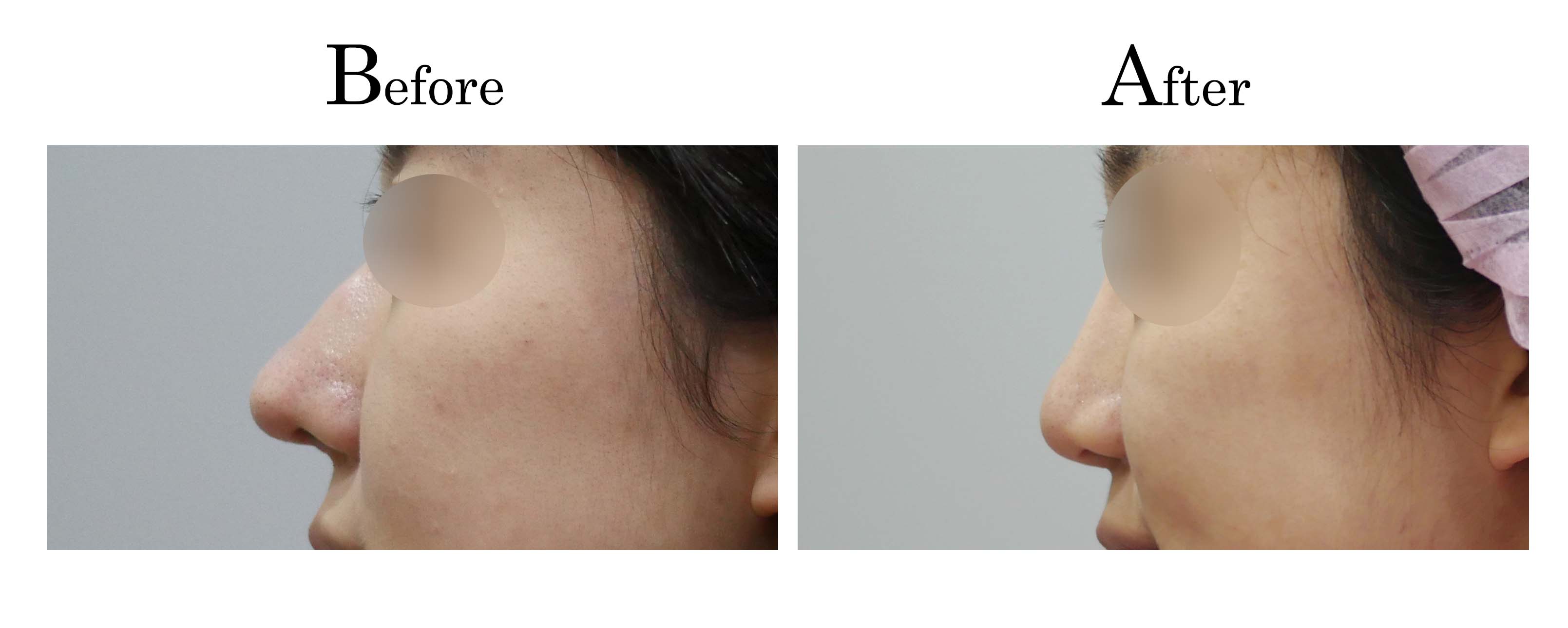
客製化鼻整形 ( Tailor-made Rhinoplasty )、微整隆鼻、結構式鼻整形 ( Structure Rhinoplasty)、功能性鼻整形、空鼻症 ( Empty Nose Syndrome)、美容性鼻整形、顏面微整形、雷射
榮譽事蹟:
“台灣首位發表"同時"進行鼻整形&鼻竇炎手術成功病例醫師” (2011)
“台灣首位發表自體軟骨鼻整形重建"小針美容"成功病例醫師”(2012)
“台灣首位發表自體軟骨鼻甲整形重建"空鼻症"成功病例醫師”(2018)
台灣“耳鼻喉頭頸外科”醫學會年會優良壁報論文 (2006)
中華民國“美容醫學”醫學會優秀論文獎 (2017)
台灣“顏面整形”重建外科醫學會年度論文獎 (2017)
台灣“顏面整形”重建外科醫學會年度SCI論文獎 (2023)
台灣“顏面整形”重建外科醫學會年度SCI論文獎 (2024)
受邀國際會議鼻整形演講:
受邀世界五大洲20多國 發表逾百場鼻整形演講及手術示範
-亞洲 : 中國、韓國、日本、泰國、新加坡、菲律賓、越南、印尼、馬來西亞、
-歐洲 : 俄羅斯, 法國, 義大利, 西班牙, 葡萄牙, 土耳其
-美洲 : 美國, 巴西, 墨西哥
-大洋洲 : 澳大利亞
-非洲 : 南非
“台灣首位受邀IFFPSS世界顏面整形大會鼻整形演講醫師”(2012 Rome)
“台灣首位連續兩年受邀AAFPRS美國顏面整形外科醫學會大會鼻整形演講醫
“台灣首位受邀IMCAS國際醫學美容抗衰老大師課程世界大會鼻整形演講醫
“台灣首位受邀IFOS世界耳鼻喉頭頸外科醫學會大會鼻整形演講醫師” (2017
“台灣首位受邀EAFPS歐洲顏面整形外科醫學會大會鼻整形演講醫師” (2017
“台灣首位受邀European ORL-HNS歐洲耳鼻喉頭頸外科醫學會大會鼻整形演講
“台灣首位受邀AAFPS澳洲顏面整形外科醫學會大會鼻整形演講醫師” (2018
“台灣首位受邀保存式鼻整形大會演講醫師” (2020 Nice)
“台灣首位受邀IFOS世界耳鼻喉頭頸外科醫學會大會發表鼻整形Keynote
Speech(長達1小時的主題演講)醫師” (2023 Dubai)
1. Advances in Facial Plastic Surgery & Rhinoplasty (Hong Kong, 2011)
2. VII International Congress – Nose & Face World (Rome,, 2012)
3. International Master Course on Aging Skin (Hong Kong, 2012)
4. 12th Annual International Rhinoplasty Course (Makati, 2013)
5. IMCAS Asia 2013 (Singapore, 2013)
6. 13th Annual International Rhinoplasty Course (Manila, 2014)
7. IMCAS Asia 2014 (Hong Kong, 2014)
8. IMCAS Asia 2015 (Bali, 2015)
9. AAFPRS fall meeting 2015 (Dallas, 2015)
10. IMCAS world congress 2016 (Paris, 2016)
11. 8th International Congress on Facial Plastic Surgery (Rio de Janeiro, 2016)
12. AAFPRS fall meeting 2016 (Nashville, 2016)
13. Pan Asia Academy of FPRS 6th Annual Congress (Manila,2016)
14. IFOS ENT World Congress 2017 (Paris, 2017)
15. 7th International Conference of PAAFPRS (Shiraz, 2017)
16. Annual Conference of EAFPS (Lisbon, 2017)
17. 4th Congress of European ORL-HNS (Barcelona, 2017)
18. 8th AFPSS Congress (Bangkok , 2017)
19. 2017 KCCS Asian Rhinoplasty Workshop (Seoul , 2017)
20. 2018 AAFPS Congress (Perth, 2018)
21. AFAS 2018 ( Seoul, 2018)
22. HSPAS 2018 International Congress (Halong, 2018)
23. IMCAS ASIA 2018 ( Bankok, 2018)
24.PAAFPRS2018(Yokohama,2018) 25.RHINOMAMA2019(Manila,2018)
26.Advancesinrhinoplasty2019(Orlando,2019)
27. 5th SRFPIC (Shiraz, 2019)
28.AMWCAsia2019(Taipei,2019)
29.Rhinoplasty Istanbul workshop (Istanbul, 2019)
30.World Laser Medicine Congress 2019 ( Taipei, 2019)
31.IMCASAsia2019(Bali,2019)
32.11DaysofFacial Plastic Surgery ( Singapore, 2019)
33.Advanced Aesthetic Rhinoplasty & Face Contouring 2019 ( St. Petersburg, 2019)
34.PAAFPRS2019(KualaLumpur,2019)
35.IMCAS world congress 2020 (Paris, 2020)
36.2ndPreservationRhinoplastyInternationalCongress(Nice,2020)
37.EAFPSAnnualMeeting2020(Virtual,2020)
38.IMCAS Asia Class (Virtual, 2020)
39.Jubilee International Scientific Conference ( St. Petersburg, 2020)
40.ENT UPDATE 2021 ( Greater Noida , 2021)
41.AMWC ASIA(Hybrid, 2021)
42.RHINOMAMA2021( Manila, 2021)
43.IMCAS ASIA 2021 ( Hybrid, 2021)
44.XIII Marius Plouznikov Memorial Lecturing ( St. Petersburg, 2021)
45.EAFPS NICE 2021 (Nice, 2021)
46. 2021 AAFPRS Annual Meeting (Virtual, 2021)
47.19th ASEAN ORL-HNS Congress (Virtual, 2021)
48.RHINOMAMA 2022 ( Manila, 2022)
49.RhinoWorld Congress 2022 ( St. Petersburg, 2022)
50.IFOS Dubai ENT World Congress 2023 ( Dubai, 2023)
51.XIInternationalInterdisciplinaryCongressofHeadandNeckDiseases(St.Petersburg,2023)
52. EAFPS Verona 2023 (Verona, 2023)
53. 8th VSAPS Annual International Scientific Congress (Ho Chi MinhCity, 2023)
54. 12th PAAFPRS International Conference (New Delhi, 2024)
55. ASEANRHINO Scientific Congress 2024 (Singapore, 2024)
56. EAFPS Istanbul 2024 (Istanbul, 2024)
57 9th HPASS Annual Scientific Conference (Hanoi City, 2024)
58. World Rhinoplasty Day (Port Elizabeth, 2024)
59. AMSC 2024 (Hong Kong, 2024)
60. Global Masters in Rhinoplasty (Rio de Janeiro, 2025)
61. 9th VSAPS Annual International Scientific Congress (Hanoi City, 2025)
【榮登國際權威】邱昱勳醫師學術成果發表於《PRS Global Open》
邱昱勳醫師長期致力於鼻整形研究,其針對「空鼻症(Empty Nose Syndrome)」的創新治療研究,於 2026 年正式發表於美國整形外科醫師協會(ASPS)官方學術期刊,象徵台灣醫療技術接軌國際。
RECONSTRUCTIVE: CASE REPORT
Empty Nose Syndrome Treated With Inferior Turbinate Augmentation Using Homologous Costal Cartilage
Chiu, Yu-Hsun MD, PhD*; Hao, Chung-Yu MD*; Kuo, Yu-Xiang MD*; Hung, Wei-Li MBBS†
Author Information
Plastic & Reconstructive Surgery-Global Open 14(2):p e7466, February 2026. | DOI: 10.1097/GOX.0000000000007466
Summary:
Empty nose syndrome (ENS) is a debilitating, iatrogenic condition caused by a previous inferior turbinectomy; the symptoms of which include subjective feelings of suffocation despite an objectively patent nasal passage. The definitive treatment for ENS is to replace the missing tissue through surgery. The implantation material and the implantation site are important considerations when it comes to ENS surgery. Here, we present a case of ENS treated with inferior turbinate augmentation using homologous costal cartilage. This novel surgical technique creates a submucosal pocket at the site of the inferior turbinate remnant and implants the homologous costal cartilage therein, thus augmenting the inferior turbinate remnant instead of creating a new turbinate. Our case reported a favorable outcome with no obvious complications. This is a rare case report of an ENS treated with inferior turbinate augmentation by using homologous costal cartilage and offers more insight into ENS surgery.
Empty nose syndrome (ENS) was first introduced in 1994 by Eugene Kern and Monika Stenkvist, who recognized it as a complication of turbinate surgery.1 It is an iatrogenic disorder defined by the presence of paradoxical nasal obstruction despite an objectively wide, patent nasal passage caused by previous inferior turbinectomy. Symptoms may include feelings of suffocation, nasal dryness, a burning sensation or pain in the nose, and nasal obstruction. Diagnosis is clinical but can be supported by results from the cotton test, the Empty Nose Syndrome 6-item Questionnaire (ENS6Q), and computed tomography imaging.1 The definitive treatment is to replace the missing tissue through surgery.
Important considerations in terms of ENS surgery include the implantation material and the implantation site. To date, multiple implantation materials have been used, and most implants are placed either on the inferior or lateral nasal wall to form a new turbinate.1,2 In the case featured here, we used a homologous costal cartilage due to its low infection rates and high biocompatibility.3 Furthermore, instead of implanting the homologous costal cartilage on the inferior or lateral nasal wall, we created a submucosal pocket at the site of the inferior turbinate and implanted the homologous costal cartilage therein. This surgical technique helps “augment” the inferior turbinate remnant instead of creating a new turbinate, and may be more physiological.
CASE REPORT
A 39-year-old man presented to our otolaryngology department with complaints of suffocation and a dry nose. According to the patient, he had undergone septomeatoplasty 19 years ago to address nasal obstruction and rhinorrhea. Since then, he had been experiencing feelings of suffocation and a dry nose, which were not relieved by conservative management, such as nasal corticosteroid sprays or oral antihistamines.
On examination, the cotton test was positive. Endoscopic findings revealed a wide-open left nasal passage with limited inferior turbinate volume (Fig. 1A). Computed tomography confirmed a severe reduction in the volume of the left inferior turbinate (Fig. 1B). The patient also scored 18 points on the self-reported ENS6Q and was subsequently diagnosed with ENS. (See table, Supplemental Digital Content 1, which displays preoperative and postoperative ENS6Q scores at 12 mo, https://links.lww.com/PRSGO/E648.)

Preoperative endoscopy findings and computer tomography. A, Preoperative endoscopic findings revealed a widely open left nasal cavity with limited left inferior turbinate volume. B, Preoperative computer tomography showed a severe reduction in left inferior turbinate volume.
Here, we introduced an innovative surgical technique involving the creation of a submucosal tunnel at the left inferior turbinate. First, cotton wools were placed anteromedially to the left resected inferior turbinate to estimate the volume required to achieve a good clinical response preoperatively. Second, under endoscopic guidance, local anesthetic was injected submucosally at the site of the left resected inferior turbinate for hydrodissection; this created a plane for our graft to be implanted and reduced the risk of perforation. Third, we dissected along the anterior portion of the remnant concha bone to create a submucosal pocket; the incision was done anteriorly, as there was more mucosa attached to the adjacent lateral wall of the nasal cavity, which could be further dissected if needed, and it allowed better surgical access. (See figure, Supplemental Digital Content 2, which displays the anterior portion of the remnant concha bone that was dissected to create a submucosal pocket. A homologous costal cartilage, carved into a fitting 3-dimensional shape according to the shape and volume of the cotton wools used, was implanted into the submucosal pocket medial to the remnant turbinate bone under endoscopic assistance, https://links.lww.com/PRSGO/E649.) If perforation were to occur, either fascia would be placed underneath the mucosa or a suture would be used to repair the perforation. Fourth, a homologous costal cartilage, carved into a fitting 3-dimensional shape according to the shape and volume of cotton wools used, was implanted into the submucosal pocket medial to the remnant turbinate bone under endoscopic assistance; medial position allowed sturdier positioning and was closer to the nasal valve, which maximized nasal resistance (Supplemental Digital Content 2, https://links.lww.com/PRSGO/E649). The homologous costal cartilage was placed such that the rounded cortical portion faced the medial border of the inferior turbinate remnant, whereas the straight cancellous portion faced the lateral wall of the nasal cavity. Finally, the incision site was sutured with a 5-0 chromic suture.
Postoperative endoscopic findings at 6 months revealed a significant increase in the volume of the left inferior turbinate (Fig. 2A). This positive outcome was further supported by the postoperative computed tomography at 6 months (Fig. 2B). The patient’s ENS6Q score, assessed 12 months after the operation, improved to 8, indicating a notable improvement compared with the preoperative score (Supplemental Digital Content 1, https://links.lww.com/PRSGO/E648).

Postoperative endoscopy findings and computer tomography. A, Six-month postoperative endoscopic findings revealed greater left inferior turbinate volume without obvious volume reduction or implant migration. B, Six-month postoperative computer tomography showed a significant increase in the volume of the neo-reconstructed left inferior turbinate after the homologous costal cartilage implantation.
DISCUSSION
When it comes to the management of ENS, conservative options include intranasal irrigation, topical ointment, antibiotic sprays, aerosols, and local corticosteroids.4 However, when conservative therapy proves to be ineffective, surgical management is recommended. Surgical management for ENS includes both injections and implants.
Injections such as hyaluronic acid, platelet-rich plasma, and lipoaspirates have been used, but their effectiveness tends to decrease after 3–6 months.5 In contrast, the effects of implants tend to be longer-lasting, and the choice of implant materials and implantation sites is an important consideration that may impact the outcome.1,2 Numerous implant materials have been used, such as autologous dermal fat and cartilages (septal, conchal, costal); homologous costal cartilage; acellular dermis graft; xenografts, such as porcine small intestine; and synthetic materials, such as polyethylene implants, silastic sheets, and beta-tricalcium phosphate implants, all of which have been shown to improve clinical outcomes.5 However, each of them has its own shortcomings. Harvesting autologous tissue creates an additional wound, which may increase postoperative pain, and site-specific complications include pneumothorax in the case of costal cartilage and limited material in the case of auricular cartilage.6 Similarly, synthetic materials have higher migration and infection rates.1 In our case, we prefer using homologous costal cartilage, which has been used previously by Malik et al7 and Chang et al8 to augment the inferior meatus in ENS surgery, with improved clinical outcomes. We prefer this material because it has advantages such as lower infection rates and higher biocompatibility when compared with synthetic implants.1,3,9 Furthermore, there was no difference in outcomes between autologous and homologous costal cartilage grafts in terms of rates of warping, resorption, infection, contour irregularity, or revision surgery when used in rhinoplasty.3
Our surgical implantation site is unconventional. Standard implantation sites include the lateral nasal wall of the inferior meatus or the inferior wall of the nasal cavity.2 A previous study has shown that lateral nasal wall implantation had better outcomes when compared with inferior wall implantation in terms of the rhinological symptoms, and it was hypothesized to be due to its more realistic representation of the missing inferior turbinate.2 In our case, we created a submucosal pocket at the site of the inferior turbinate and implanted the homologous costal cartilage therein, thus recreating the original resected inferior turbinate through augmentation. This is different from inferior meatus augmentation, where the incision is made more anteriorly at the limen nasi, and a neoturbinate is formed more inferiorly at the inferior meatus.7,8 Inferior turbinate augmentation is more physiological and has achieved favorable outcomes. However, it is surgically more demanding and requires the presence of an inferior turbinate remnant. This is a rare case report of an ENS treated with inferior turbinate augmentation using homologous costal cartilage and offers further insight into ENS surgery.
DISCLOSURE
The authors have no financial interest to declare in relation to the content of this article.
PATIENT CONSENT
Written informed consent was obtained from the legally authorized representative(s) for the anonymized patient information to be published in this article.
ACKNOWLEDGMENTS
Ethical approval to report this case was obtained from the institutional review board of Shin Kong Wu Ho-Su Memorial Hospital (approval ID: 20230501R). All procedures in this study were conducted in accordance with the institutional review board.
REFERENCES
1. Talmadge J, Nayak JV, Yao W, et al. Management of postsurgical empty nose syndrome. Facial Plast Surg Clin North Am. 2019;27:465–475.
• Cited Here |
• Google Scholar
2. Lee TJ, Fu CH, Wu CL, et al. Surgical outcome for empty nose syndrome: Impact of implantation site. Laryngoscope. 2018;128:554–559.
• Cited Here |
• Google Scholar
3. Vila PM, Jeanpierre LM, Rizzi CJ, et al. Comparison of autologous vs homologous costal cartilage grafts in dorsal augmentation rhinoplasty. JAMA Otolaryngol Head Neck Surg. 2020;146:347–354.
• Cited Here |
• Google Scholar
4. Saafan ME, Hegazy HM, Albirmawy OA. Empty nose syndrome: etiopathogenesis and management. Egypt J Otolaryngol. 2016;32:119–129.
• Cited Here |
• Google Scholar
5. Kim DH, Basurrah MA, Kim SW, et al. Surgical and regenerative treatment options for empty nose syndrome: a systematic review. Clin Exp Otorhinolaryngol. 2024;17:241–252.
• Cited Here |
• Google Scholar
6. Hosokawa Y, Miyawaki T, Omura K, et al. Surgical treatment for empty nose syndrome using autologous dermal fat: evaluation of symptomatic improvement. Ear Nose Throat J. 2022;104:1455613221130885.
• Cited Here |
• Google Scholar
7. Malik J, Dholakia S, Spector BM, et al. Inferior meatus augmentation procedure (IMAP) normalizes nasal airflow patterns in empty nose syndrome patients via computational fluid dynamics (CFD) modeling. Int Forum Allergy Rhinol. 2021;11:902–909.
• Cited Here |
• Google Scholar
8. Chang MT, Bartho M, Kim D, et al. Inferior meatus augmentation procedure (IMAP) for treatment of empty nose syndrome. Laryngoscope. 2022;132:1285–1288.
• Cited Here |
• Google Scholar
9. Hu HC, Fang HW, Chiu YH. Delayed-onset edematous foreign body granulomas 40 years after augmentation rhinoplasty by silicone implant combined with liquid silicone injection. Aesthetic Plast Surg. 2017;41:637–640.
• Cited Here |
• Google Scholar